Fantastic Fluids

Introduction to the Unit
The study of fluids, which can be either liquids or gases, introduces students to fluid mechanics, an area of knowledge important in many industries (such as aeronautics, engineering, meteorology, and oceanography). Fluids, including air and water, are essential to many industrial processes and form the basis of hydraulic and pneumatic devices. Students will learn about the properties of fluids by experimenting with and investigating the viscosity and density of different liquids and ways in which these properties affect objects placed in those liquids. Students will explore the implications of Archimedes’ Principle by investigating and measuring the buoyant forces on different objects. As well, they will learn about the diverse applications of the principles involved in fluid mechanics, including industrial applications such as jet propulsion, and everyday applications such as ensuring that sauces are cooked to the right consistency (degree of viscosity).
The study of fluids, which can be either liquids or gases, introduces students to fluid mechanics, an area of knowledge important in many industries (such as aeronautics, engineering, meteorology, and oceanography). Fluids, including air and water, are essential to many industrial processes and form the basis of hydraulic and pneumatic devices. Students will learn about the properties of fluids by experimenting with and investigating the viscosity and density of different liquids and ways in which these properties affect objects placed in those liquids. Students will explore the implications of Archimedes’ Principle by investigating and measuring the buoyant forces on different objects. As well, they will learn about the diverse applications of the principles involved in fluid mechanics, including industrial applications such as jet propulsion, and everyday applications such as ensuring that sauces are cooked to the right consistency (degree of viscosity).
Fluids

Remember:
A Fluid is a state of matter, such as liquid or gas, in which the component particles (generally molecules) can move past one another. Fluids flow easily and conform to the shape of their containers. Solids are not considered to be fluids because their molecules cannot move past one another and thus they have a definite shape.
Viscosity refers to the resistance of a substance to flow. For example, water has a lower viscosity than molasses and flows more easily.
Flow Rate is the the amount of fluid that flows in a given time across a given distance. Flow rate provides a measurement to determine how viscous (thick or thin) a fluid is. The viscosity (and thus flow rate) of a fluid can be altered by temperature.
A Fluid is a state of matter, such as liquid or gas, in which the component particles (generally molecules) can move past one another. Fluids flow easily and conform to the shape of their containers. Solids are not considered to be fluids because their molecules cannot move past one another and thus they have a definite shape.
Viscosity refers to the resistance of a substance to flow. For example, water has a lower viscosity than molasses and flows more easily.
Flow Rate is the the amount of fluid that flows in a given time across a given distance. Flow rate provides a measurement to determine how viscous (thick or thin) a fluid is. The viscosity (and thus flow rate) of a fluid can be altered by temperature.
State of Matter
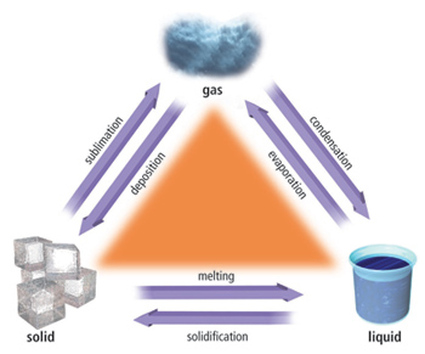
Reminders:
Change of State - The transformation of a substances physical state into another state.
Melting - The process of changing from a solid to a liquid.
Vaporization - The process of changing from a liquid to gas.
Condensation - The process of changing from a gas to a liquid.
Freezing - The process of changing from a liquid to a solid.
Sublimation - The process of changing from a solid to a gas without going through the liquid state.
Change of State - The transformation of a substances physical state into another state.
Melting - The process of changing from a solid to a liquid.
Vaporization - The process of changing from a liquid to gas.
Condensation - The process of changing from a gas to a liquid.
Freezing - The process of changing from a liquid to a solid.
Sublimation - The process of changing from a solid to a gas without going through the liquid state.
Short Video on States of Matter
Please complete page 257 and page 258
| science8_page_257.pdf | |
| File Size: | 52 kb |
| File Type: | |
| science_page_258.pdf | |
| File Size: | 45 kb |
| File Type: | |
In small groups, the students created slime also known as "GLU" in French and got the opportunity to play with it and watch its magical properties. To recreate the Glu at home see the following informational sheets.
In small groups, the students created oobleck and got the opportunity to play with it and watch its magical properties.
To find out more information about suspension, and how to create this easy mixture at home, visit the following webpage http://bit.ly/nA3yRf
Watch the following clip to see a humorous (but educational) experience Ellen Degeneres had with magic mud on her show. You'll literally see someone walk on water:
WHY VISCOSITY VARIES

Factors That Affect Flow Rate
(1) Temperature has an affect on flow rate. In most cases, the hotter the temperature the higher the flow rate, while the colder the temperature the slower the flow rate.
(2) Fluids have different viscosities and size of the particles (bulkiness).
(3) Internal friction (rubbing) of particles as they slip past each other also varies between liquids.
Today students will watch a short video on "The Boston Molasses Disaster of 1919." This documentary (created by the History Channel - Biggest Disasters of the 20th Century) not only describes the tragic impact that temperature can have on fluids, but more importantly demonstrates the need for humans to not cut corner when constructing tanks that hold large quantities of certain fluids.
The following two clips provide a description of the events leading up to, during and then after this disastrous flood.
The Boston Molasses Disaster of 1919


